Popular on Rezul
- Still Using Ice? FrostSkin Reinvents Hydration - 131
- Ice Melts. Infrastructure Fails. What Happens to Clean Water?
- Mend Colorado Launches Revamped Sports Performance Training Page
- Cold. Clean. Anywhere. Meet FrostSkin
- Actor, Spokesperson Rio Rocket Featured in "Switch to AT&T" Campaign Showing How Customers Can BYOD and Keep Their Number
- Novestco Revolutionizes Deal Screening, Delivering Comprehensive Feasibility Analysis in Minutes
- The World's No.1 Superstar® Brings Disco Fever Back With New Global Single and Video "Disco Dancing"
- Boston Industrial Solutions' Natron® 512N Series UV LED Ink Achieves BPA Certification, Advancing Safe and Sustainable Digital Printing
- New Children's Picture Book "Diwa of Mount Luntian" Focuses on Calm, Culture, and Connection for Today's Families
- Work 365 Delivers Purpose-Built Revenue Operations for Microsoft Cloud for US Government
Similar on Rezul
- Strong Clinical Results for Breakthrough Liver Diagnostic Platform; ENDRA Life Sciences (N A S D A Q: NDRA) $NDRA
- Phoenix Rebellion Therapy Celebrates 10 Years Helping Utahns Overcome Trauma as Utah Faces Nation's 2nd-Highest Rate of Mental Health Challenges
- Notice: Hrm Queen Laurence I Assumes Crown Control & $317q Fund. 3bn Unopoly Shares Settled. Requisition Of Buckingham Palace & Windsor Castle Final
- Yoga Retreats, Ecstatic Dance & Spiritual App launched
- Elder Abuse Case Against Healthy Traditions Owner Raises Questions As To The Dire Reality Of Abuse Against The Last Of The Baby Boomers
- Claude Riveloux Review 2026: How the $10B Fund Manager Dispels 'Scam' Rumors Through Education
- Dr. Sheel Desai Solomon and Preston Dermatology Continue Awards Streak with Top Honors in 2026 Maggy Awards
- $167 Billion Pharma R&D Market Largely Untapped by AI Creates Major Growth Runway for KALA Bios Data-Sovereign AI Strategy: N A S D A Q: KALA
- Memelinked Social Media powered by cryptocurrency launching July 2026
- Progressive Dental & The Closing Institute Partner with Zest Dental Solutions to Elevate Full-Arch Growth and Patient Outcomes
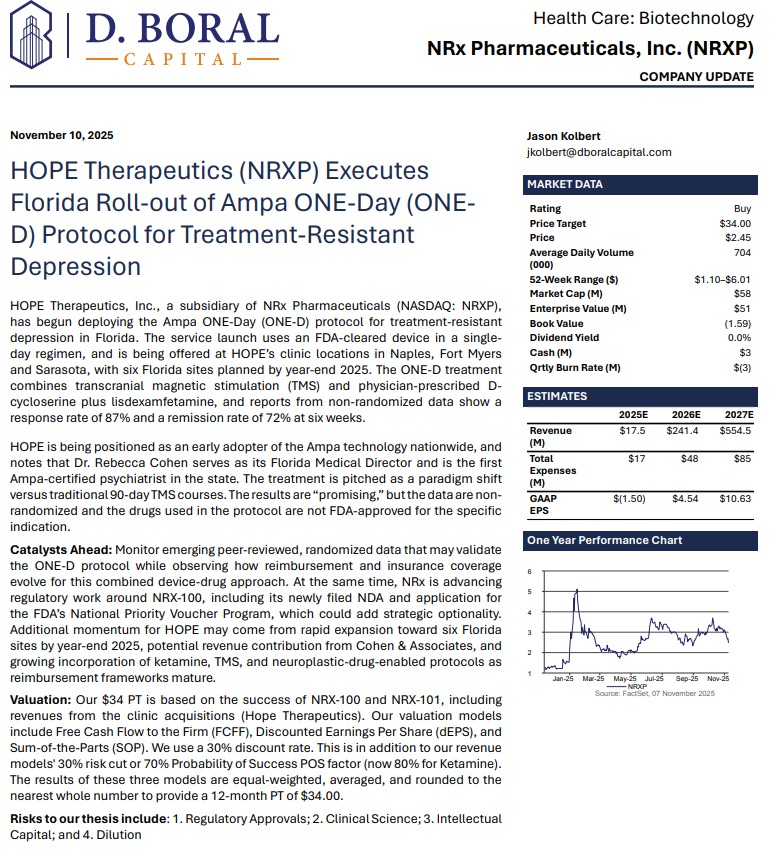
High-Conviction CNS Disruptor Aiming to Transform Suicidal Depression, Ketamine Therapeutics, and TMS - Reaching Millions by 2030
Rezul News/10721927
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) NRXP Also Reports Its Superior Preservative-Free IV Ketamine Now Submitted for FDA Abbreviated New Drug Application
MIAMI - Rezul -- In a mental-health landscape where more than 13 million Americans seriously consider suicide each year, few companies are positioned as boldly—or as comprehensively—as NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP). With a newly expanded pipeline, a validated FDA regulatory path, revenue-generating clinic operations, and a third-party $34 analyst price target, NRx is surfacing as one of the most disruptive stories in central nervous system (CNS) therapeutics.
Today, the company is advancing three strategic pillars with the potential to reshape treatment for suicidal depression, chronic pain, and adjunctive neuromodulation:
Layered with manufacturing readiness, clinical-care expansion, and secured operating capital through July 2026, NRXP enters 2026 positioned for clinical, regulatory, and commercial convergence.
A Potential Million-Patient Market by 2030: The New NRX-101 TMS Indication
One of the most noteworthy catalysts for NRXP emerged in Q4 when the company amended its IND for NRX-101 to include use alongside Transcranial Magnetic Stimulation (TMS).
This is not incremental—it's transformational.
TMS is experiencing rapid adoption, with projections indicating over one million Americans may receive TMS annually by 2030. Yet, recent data indicate that combining TMS with NMDA-modulating therapeutics may dramatically enhance patient outcomes.
In newly presented Real World Data using a modern Theta Burst TMS device and single-day treatment protocol:
NRX-101 is uniquely positioned for this indication because:
NRXP anticipates that a confirmatory ~120-patient trial could support FDA registration for the TMS augmentation indication. Partnership discussions are already underway with TMS device manufacturers to co-develop the pivotal trial and pursue joint labeling.
More on Rezul News
For investors, this opens access to a new multi-billion-dollar neuromodulation-pharma hybrid market that previously did not exist.
KETAFREE™: A Clean-Label Ketamine Positioned to Redefine a $750 Million Market
On December 2, NRXP announced a milestone with major commercial implications:
The FDA has officially received and validated the company's ANDA for KETAFREE™, its preservative-free IV ketamine formulation.
The ANDA has been deemed "substantially complete" and assigned a GDUFA goal date of July 29, 2026.
Why this matters:
1. The First Preservative-Free Ketamine in the U.S.
Most ketamine products contain benzethonium chloride (BZT)—a preservative not recognized as safe by the FDA and banned in topical antiseptics.
NRXP has filed a Citizen Petition requesting the FDA remove BZT from all U.S. ketamine products.
2. A $750M Global Generic Market Ready for Disruption
KETAFREE™ targets all existing ketamine indications with a cleaner safety profile and U.S.-based manufacturing.
The company has already manufactured initial registration lots and is prepared to scale to one million vials per month.
3. A Strategic Complement to NRX-100
KETAFREE™ follows the generic regulatory pathway (ANDA), while NRX-100 follows the innovative (NDA) pathway for suicidal depression and carries FDA Fast Track designation.
This dual-path approach not only expands the addressable market—it derisks commercialization.
NRX-100 (IV Ketamine): Fast Track, Real-World Evidence, and a Race to Fill an Unmet Clinical Void
NRXP continues rapid progress on NRX-100, an innovative ketamine-based therapy specifically developed for acute suicidal ideation.
Key elements:
This stands in contrast to Spravato®, which despite expected $1.6 billion in 2025 sales, carries labeling stating it has not been demonstrated to reduce suicidal ideation or prevent suicide.
NRXP is also seeking a Commissioner's National Priority Voucher, which could accelerate review even further.
HOPE Clinics: A Rapid-Growing Revenue Engine
More on Rezul News
2025 marked NRXP's entry into active revenue generation through its HOPE Therapeutics subsidiary clinics.
This expansion supports near-term operating revenue while building a deployment platform for NRX-100 and KETAFREE™ upon approval.
Corporate Strength: Funding, Manufacturing, and Operational Execution
NRXP has accomplished several critical operational milestones:
• Cash runway secured through July 2026
Providing stability to complete pivotal regulatory steps.
• Multiple commercial drug lots manufactured
With stability data supporting three-year room-temperature shelf life.
• Real-World Data validating NRX-101's TMS synergy
Creating a new, fast-to-market indication.
• FDA Suitability Petition granted
Confirming a safe regulatory path for preservative-free ketamine.
• Analyst Price Target: $34
In an independent report by D. Boral.
Why Investors Are Paying Attention
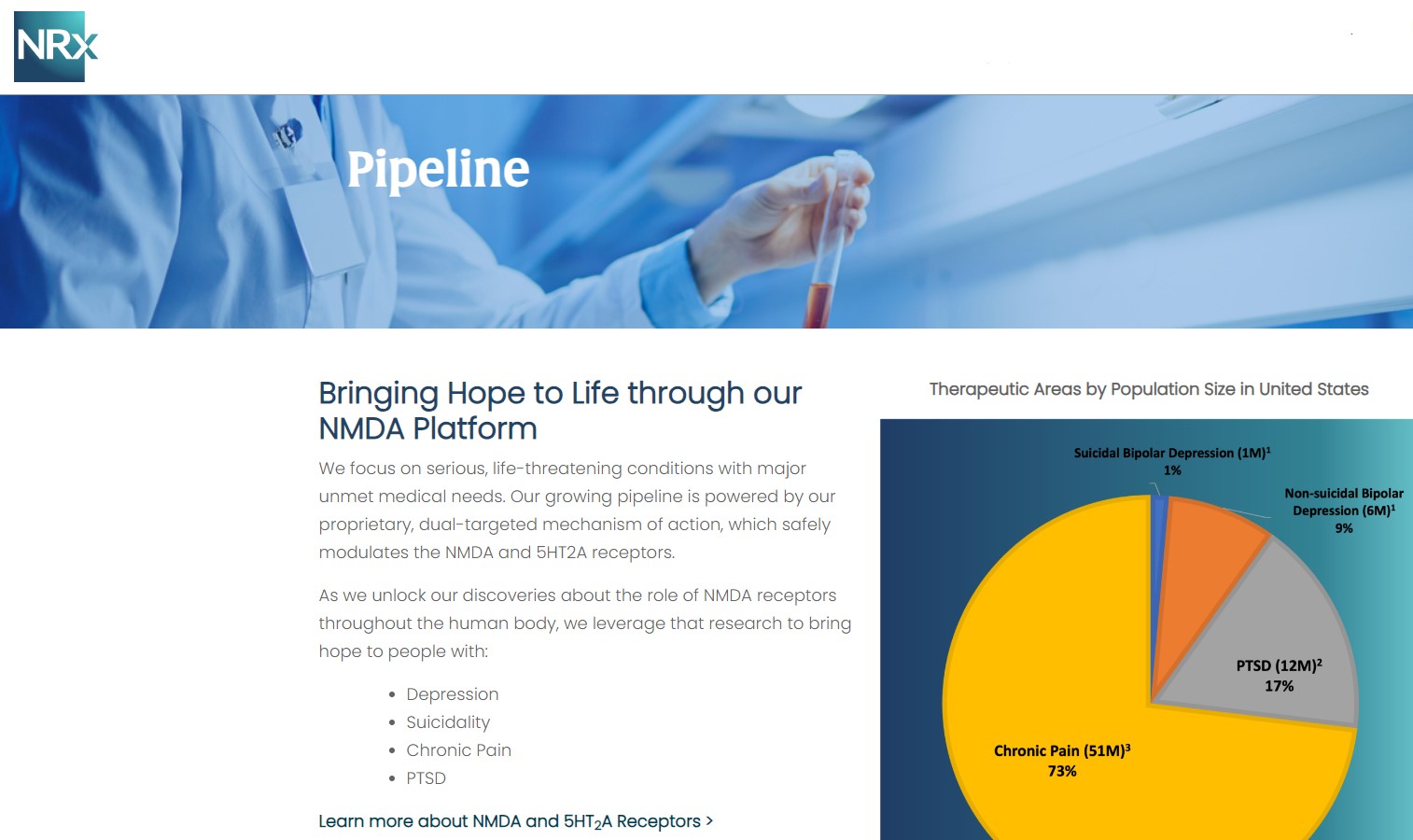
NRx Pharmaceuticals is not a single-asset story. It is a converging portfolio of:
With suicide now a top public-health priority, NRXP is aligning itself at the crossroad of clinical need, regulatory urgency, and market expansion.
The Bottom Line
NRx Pharmaceuticals is shaping up to be one of the most compelling CNS-focused companies entering 2026. By simultaneously innovating, commercializing, and scaling, NRXP is positioning itself to influence multiple high-value markets—from TMS augmentation to suicidal depression to chronic pain and preservative-free ketamine.
As the company's clinical, regulatory, and commercial inflection points approach, NRXP is becoming one of the most closely watched emerging players in neuropsychiatry and mental-health therapeutics.
For more information:
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Website: www.nrxpharma.com
Media Contact: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com | Phone: (484) 254-6134
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Today, the company is advancing three strategic pillars with the potential to reshape treatment for suicidal depression, chronic pain, and adjunctive neuromodulation:
- NRX-101 – an FDA Breakthrough Therapy now targeting a newly emerging market in TMS augmentation.
- NRX-100 (IV ketamine) – an innovative therapy for acute suicidality under FDA Fast Track designation.
- KETAFREE™ – the first preservative-free IV ketamine to reach FDA ANDA review.
Layered with manufacturing readiness, clinical-care expansion, and secured operating capital through July 2026, NRXP enters 2026 positioned for clinical, regulatory, and commercial convergence.
A Potential Million-Patient Market by 2030: The New NRX-101 TMS Indication
One of the most noteworthy catalysts for NRXP emerged in Q4 when the company amended its IND for NRX-101 to include use alongside Transcranial Magnetic Stimulation (TMS).
This is not incremental—it's transformational.
TMS is experiencing rapid adoption, with projections indicating over one million Americans may receive TMS annually by 2030. Yet, recent data indicate that combining TMS with NMDA-modulating therapeutics may dramatically enhance patient outcomes.
In newly presented Real World Data using a modern Theta Burst TMS device and single-day treatment protocol:
- 87% of patients achieved clinical response
- 72% reached remission at 6 weeks
These results are particularly striking given a single administration of oral D-cycloserine, a key component of NRX-101.
NRX-101 is uniquely positioned for this indication because:
- It contains lurasidone, counteracting the low-grade hallucination risk of D-cycloserine alone.
- It holds composition-of-matter patent protection globally.
- It is an FDA Breakthrough Therapy, enabling expedited development and review.
NRXP anticipates that a confirmatory ~120-patient trial could support FDA registration for the TMS augmentation indication. Partnership discussions are already underway with TMS device manufacturers to co-develop the pivotal trial and pursue joint labeling.
More on Rezul News
- Strong Clinical Results for Breakthrough Liver Diagnostic Platform; ENDRA Life Sciences (N A S D A Q: NDRA) $NDRA
- 46th International Symposium On Forecasting – Dates, Venue And Speakers Announced
- Phoenix Rebellion Therapy Celebrates 10 Years Helping Utahns Overcome Trauma as Utah Faces Nation's 2nd-Highest Rate of Mental Health Challenges
- Bonavita Luxury & Portable Lavatories Announces Rebrand to Bonavita Site Solutions
- Raleigh Emerges as a Key Player in Sustainable Fashion Innovation for 2026
For investors, this opens access to a new multi-billion-dollar neuromodulation-pharma hybrid market that previously did not exist.
KETAFREE™: A Clean-Label Ketamine Positioned to Redefine a $750 Million Market
On December 2, NRXP announced a milestone with major commercial implications:
The FDA has officially received and validated the company's ANDA for KETAFREE™, its preservative-free IV ketamine formulation.
The ANDA has been deemed "substantially complete" and assigned a GDUFA goal date of July 29, 2026.
Why this matters:
1. The First Preservative-Free Ketamine in the U.S.
Most ketamine products contain benzethonium chloride (BZT)—a preservative not recognized as safe by the FDA and banned in topical antiseptics.
NRXP has filed a Citizen Petition requesting the FDA remove BZT from all U.S. ketamine products.
2. A $750M Global Generic Market Ready for Disruption
KETAFREE™ targets all existing ketamine indications with a cleaner safety profile and U.S.-based manufacturing.
The company has already manufactured initial registration lots and is prepared to scale to one million vials per month.
3. A Strategic Complement to NRX-100
KETAFREE™ follows the generic regulatory pathway (ANDA), while NRX-100 follows the innovative (NDA) pathway for suicidal depression and carries FDA Fast Track designation.
This dual-path approach not only expands the addressable market—it derisks commercialization.
NRX-100 (IV Ketamine): Fast Track, Real-World Evidence, and a Race to Fill an Unmet Clinical Void
NRXP continues rapid progress on NRX-100, an innovative ketamine-based therapy specifically developed for acute suicidal ideation.
Key elements:
- Fast Track Designation from the FDA.
- An NDA expected to be completed in Q4 2025.
- Inclusion of real-world outcomes from 60,000+ IV ketamine patients, compared with 6,000 intranasal S-ketamine patients.
- Interim data from 20,000 IV ketamine patients show a faster onset and greater effect size relative to nasal S-ketamine.
This stands in contrast to Spravato®, which despite expected $1.6 billion in 2025 sales, carries labeling stating it has not been demonstrated to reduce suicidal ideation or prevent suicide.
NRXP is also seeking a Commissioner's National Priority Voucher, which could accelerate review even further.
HOPE Clinics: A Rapid-Growing Revenue Engine
More on Rezul News
- Notice: Hrm Queen Laurence I Assumes Crown Control & $317q Fund. 3bn Unopoly Shares Settled. Requisition Of Buckingham Palace & Windsor Castle Final
- 13 Full Moons of Black Dandelion Convergent Voice™ An Integration of Literacy & Wellness Symposium
- Yoga Retreats, Ecstatic Dance & Spiritual App launched
- Elder Abuse Case Against Healthy Traditions Owner Raises Questions As To The Dire Reality Of Abuse Against The Last Of The Baby Boomers
- JiT Home Buyers Explains the 70% Rule Investors Use When Evaluating Residential Properties
2025 marked NRXP's entry into active revenue generation through its HOPE Therapeutics subsidiary clinics.
- Three facilities currently operating in Florida.
- Three additional locations expected by year-end.
- Focus includes depression, PTSD, interventional psychiatry, military and first-responder mental-health care.
This expansion supports near-term operating revenue while building a deployment platform for NRX-100 and KETAFREE™ upon approval.
Corporate Strength: Funding, Manufacturing, and Operational Execution
NRXP has accomplished several critical operational milestones:
• Cash runway secured through July 2026
Providing stability to complete pivotal regulatory steps.
• Multiple commercial drug lots manufactured
With stability data supporting three-year room-temperature shelf life.
• Real-World Data validating NRX-101's TMS synergy
Creating a new, fast-to-market indication.
• FDA Suitability Petition granted
Confirming a safe regulatory path for preservative-free ketamine.
• Analyst Price Target: $34
In an independent report by D. Boral.
Why Investors Are Paying Attention
NRx Pharmaceuticals is not a single-asset story. It is a converging portfolio of:
- Breakthrough-designated CNS drugs
- A first-in-class preservative-free ketamine
- A new TMS-augmentation market with million-patient potential
- A scalable manufacturing base
- A growing national clinical footprint
- A strategic partnership with Alvogen
- Fast Track regulatory momentum
With suicide now a top public-health priority, NRXP is aligning itself at the crossroad of clinical need, regulatory urgency, and market expansion.
The Bottom Line
NRx Pharmaceuticals is shaping up to be one of the most compelling CNS-focused companies entering 2026. By simultaneously innovating, commercializing, and scaling, NRXP is positioning itself to influence multiple high-value markets—from TMS augmentation to suicidal depression to chronic pain and preservative-free ketamine.
As the company's clinical, regulatory, and commercial inflection points approach, NRXP is becoming one of the most closely watched emerging players in neuropsychiatry and mental-health therapeutics.
For more information:
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Website: www.nrxpharma.com
Media Contact: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com | Phone: (484) 254-6134
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
0 Comments
Latest on Rezul News
- Dr. Sheel Desai Solomon and Preston Dermatology Continue Awards Streak with Top Honors in 2026 Maggy Awards
- JiT Home Buyers Discusses Growing Challenges of Vacant Homes Across U.S. Housing Markets
- Jack and Sage Acquires Sustainable Apparel Brand Kastlfel, Expanding Premium Logo Wear Across National Parks and Ski Resorts
- Green Roofing Options Growing in Popularity Across Oklahoma
- Turnleaf opens seven new decorated model homes
- Alpine Building Performance Launches First Free AI Inspection Forecaster for Real Estate Agents
- Greater Houston Houses LLC Celebrates 20 Years of Helping Houston Homeowners Sell Fast
- Vicinity Disrupts the Lead-Gen Status Quo:Launches Subscription-Based "Ecosystem" with Zero Referral
- Drew Davis Joins Berkshire Hathaway HomeServices Hilton Head Bluffton Realty
- Cancun International Airport Prepares for Record Travel Surge Ahead of Spring Break, Summer, and the 2026 High Season
- $167 Billion Pharma R&D Market Largely Untapped by AI Creates Major Growth Runway for KALA Bios Data-Sovereign AI Strategy: N A S D A Q: KALA
- Lighthouse Tech Awards Recognize Top HR Technology Providers for 2026
- ADB Selects OneVizion to Advance Field Execution and Infrastructure Program Management
- Memelinked Social Media powered by cryptocurrency launching July 2026
- Colliers represents seller in sale of 34,430-square-foot office building in Sugar Land, Texas
- Maui Luxury Real Estate Agents Share Fun Ways to Stay Active on Maui!
- Seven-Year-Old Toronto Dancer Julianna Selivanov Wins Nine Medals at Quebec Championship and Reaches Finals at UK Dance Festival
- PulteGroup expands Northeast Florida presence with Seminole Palms and Lakeview Estates
- JiT Home Buyers Provides Guidance for Families Navigating Inherited Homes Nationwide
- Progressive Dental & The Closing Institute Partner with Zest Dental Solutions to Elevate Full-Arch Growth and Patient Outcomes